Doctors Trust
Town & Country Compounding
for high-quality compounded medicine that makes patients better.

Doctors Trust
Town & Country Compounding
for high-quality compounded medicine that makes patients better.
top 1% of compounding pharmacies
Our accreditations place us in the top 1% of compounding pharmacies
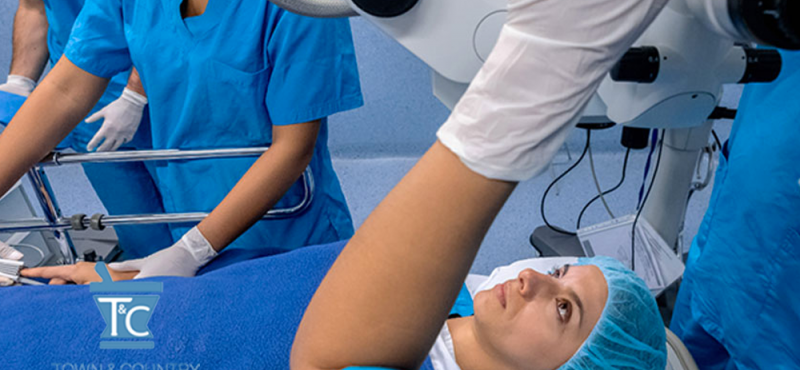
Do you know how your compounded medicines are made?
Let us show you how we make ours.


Take a tour of our high-tech sterile and non-sterile compounding labs.
Rigorous protocols lead to excellent patient outcomes.
Practitioners see the difference
Town & Country Compounding is head and shoulders above the rest.
Town & Country Compounding's LDN titration kits have helped my success rates in managing pain in patients with autoimmune diseases.

Hackensack, NJ
Town & Country Compounding makes customized medication at the highest level.
Their high-tech labs, expertise, and rigorous compounding protocols give me confidence that my patients are getting the best quality compounds.

Bryn Mawr, PA
I recommend Town & Country Compounding to all my patients.
Their facility is top-notch. They’ve got a beautiful new lab. You can rely on their compounded medicine.

Franklin Lakes, NJ
Town & Country Compounding’s commitment to quality is the best in the area.
I checked out the processes at their lab and was impressed. Patients feel better when they use medicine from Town & Country.

Ridgewood, NJ
Patients do better when they get their medicine from Town & Country Compounding.
They’ve invested in quality assurance - that’s what you’re paying for. I rely on Town & Country as our go-to compounding pharmacy.

Harrington Park, NJ
For the past 25 years, I’ve guided my patients to use Town & Country Compounding.
We have remained loyal because
Town & Country enables me to manage my patients‘ pain with confidence that their medicines are at the highest quality.

Hackensack, NJ
Why it’s important to choose the right compounding pharmacy
Where you get your medicine compounded makes a big difference in your health outcomes.
Not all pharmacies uphold the same standards.
Unlike a commercial drug you get at a retail pharmacy that will have the same quality no matter where you buy it, the effectiveness of compounded medicine can vary widely from pharmacy to pharmacy.
Town & Country Compounding makes customized medicine to the highest standards.
Compounding Specialties
Learn about our high-quality compounded products
Satisfied patients feel the difference
I don’t know what we would do without them. They are experts and extremely knowledgeable with complex compounding requests. They have always been patient with my questions and comments, and they work seamlessly with my doctor regarding refills and changes. They have my trust, and my thanks.
The staff is very efficient and the quality of the pharmaceutical product is superior. Months ago I switched to a local compounding pharmacy and got into trouble because of the inferiority of their product. I went back to Town & Country immediately and have been with them ever since!
I had a nightmarishly convoluted problem with one of my prescriptions. Ann not only untangled the error, but also: 1. Emailed me an actual calendar to illustrate the correct schedule for my refills. 2. Educated me on multiple points regarding the medication I take (I'm just a regular person and the information she gave me will be incredibly useful going forward). 3. Created a solution to my shipping dilemma. 4. Communicated back and forth with my doctor throughout the entire process. I have never experienced this quality of service, this level of professionalism, and this caliber of work. Thank you, Ann.
Town & Country Compounding recently moved to update their facilities and their staff remains top-notch. I trust them with all my compounding needs and have already referred my friends to mention Town & Country to their doctors if they need compounded/custom medicine.
I don’t know what we would do without them. They are experts and extremely knowledgeable with complex compounding requests. They have always been patient with my questions and comments, and they work seamlessly with my doctor regarding refills and changes. They have my trust, and my thanks.
The staff is very efficient and the quality of the pharmaceutical product is superior. Months ago I switched to a local compounding pharmacy and got into trouble because of the inferiority of their product. I went back to Town & Country immediately and have been with them ever since!
I had a nightmarishly convoluted problem with one of my prescriptions. Ann not only untangled the error, but also: 1. Emailed me an actual calendar to illustrate the correct schedule for my refills. 2. Educated me on multiple points regarding the medication I take (I'm just a regular person and the information she gave me will be incredibly useful going forward). 3. Created a solution to my shipping dilemma. 4. Communicated back and forth with my doctor throughout the entire process. I have never experienced this quality of service, this level of professionalism, and this caliber of work. Thank you, Ann.
Town & Country Compounding recently moved to update their facilities and their staff remains top-notch. I trust them with all my compounding needs and have already referred my friends to mention Town & Country to their doctors if they need compounded/custom medicine.