Did you know that Town & Country Compounding helps with the development of new drugs?
We take pride in working with researchers from initial development of the study or trial until completion.
Preclinical research and clinical trials are a necessity for new drug development. Town & Country Compounding Pharmacy has had considerable experience working with pharmaceutical companies, universities, hospitals and physician groups to meet their needs, especially in early phase drug development. Our team often assists the researcher or drug company with suggestions to the development of the study plan and/or research protocol before the clinical trial begins.
Sometimes our clinical research clients need only ten doses. Sometimes they need thousands of doses.
Whatever the need, Town & Country Compounding can deliver!
We recently relocated to expand our operations and increase our capabilities for compounding for clinical trials and clinical research.






State-of-the-Art Laboratories & Cleanrooms
Our larger labs and cleanrooms were built with the highest standards required by the FDA and the State Board of Pharmacy.
Town & Country Compounding has participated in studies and clinical trials for many types of active ingredients, combination drugs, and various dosage forms including but not limited to:
- Sterile drugs used in IV’s (parenteral medications)
- Vials
- IV bags
- Prefilled syringes
- Alternative bases for drugs to achieve better active drug delivery
- Oral medications
- Delayed release capsules
- Immediate release capsules
- Sterile ophthalmic drugs
- Sterile bladder irrigations
- Nasal sprays and irrigations
- Topical creams and gels


Drug Development
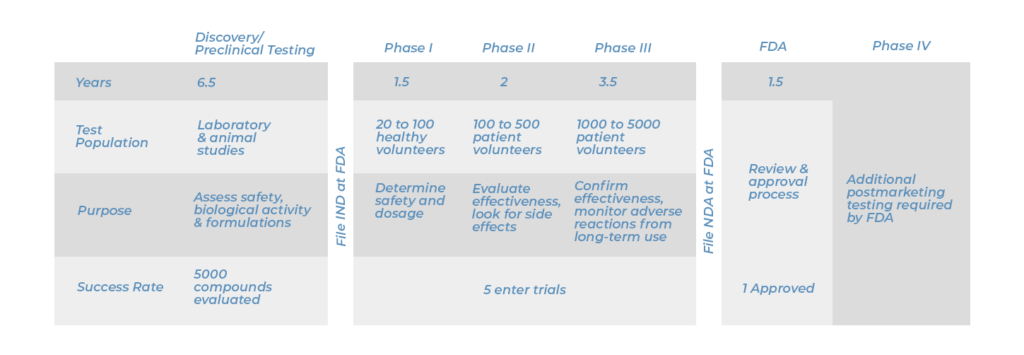
It can take 12-15 years from when a drug gets developed to when (or if) it makes it to become an FDA approved drug on the market. Drug development is very complex. Actually, only a few of the drugs (5 out of 5000 as shown in the chart) that are developed even make it through to the point of being tested on humans (Phase I below), and out of 5000 drugs discovered, only 1 makes it through for FDA approval.
A study published in March 2020 estimates that the median cost to bring a new drug to market is $985 million.
Clinical Trials Have Five Phases
Only after preclinical research, if a new treatment has the potential to help patients, may it continue to a clinical trial.
- Phase 0: Investigative phase; there are very few human participants who get the drug, and microdosing is used
- Nontoxic doses are given to a very small group of patients for only a short period of time. For example, this phase is considered beneficial for patients with aggressive brain tumors called glioblastomas.


- Phase 1: A small number of healthy volunteers participate, and the studies focus on safety.
- Phase 2: Preliminary data on effectiveness of drug is studied. Participants receiving the drug may be compared with similar participants receiving a placebo. We ask: Are there short-term side effects?
- Phase 3: Additional information about safety and effectiveness is studied. Different dosages, different populations, and combinations with other drugs are studied.
- Phase 4: Studies that gather additional information about a drug’s safety, efficacy, or ideal use that are required after a drug has been approved and is on the market.
Source: NCCN ClinicalTrials.org
Discover how Town & Country Compounding contributes to the critical phases of clinical trials, from formulation development to stability testing, and how these efforts support groundbreaking medical advancements.
Whether you’re involved with clinical trials (within USA or International), this video will provide you with a unique perspective on the challenges and innovations in the field.
Let’s Get Started
We take pride in working with your team from initial development of the study or trial and through all aspects of the study or trial up until completion.
References
- https://www.healio.com/news/hematology-oncology/20190410/phase-0-trials-quickest-route-for-development-of-glioblastoma-treatments
- https://www.nccn.org/patients/resources/clinical_trials/phases.aspx
- https://www.clinicaltrials.gov/ct2/help/glossary/phase
- https://www.fda.gov/media/82381/download